 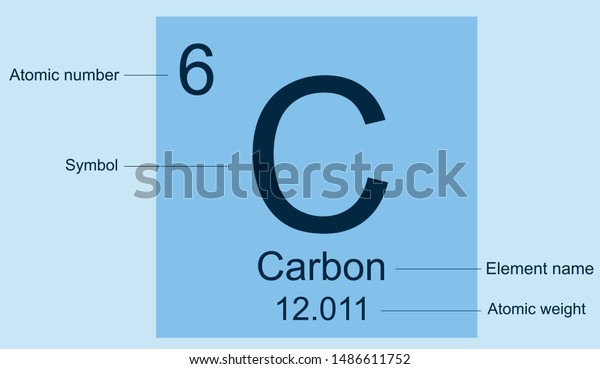
That means that all sodium atoms have 11 protons. For example, the atomic number (z) for sodium (Na) is 11. The symbol for the atomic number is designated with the letter Z. This number is known as the atomic number, which identifies the number of protons in the nucleus of ALL atoms in a given element. When you study the periodic table, the first thing that you may notice is the number that lies above the symbol. Carbon-14, an isotope with a half-life of 5715 years, has been widely used to date such materials as wood, archaeological specimens, etc.\) In 1961 the International Union of Pure and Applied Chemistry adopted the isotope carbon-12 as the basis for atomic weights. Some of the most important compounds of carbon are carbon dioxide (CO 2), carbon monoxide (CO), carbon disulfide (CS 2), chloroform (CHCl 3), carbon tetrachloride (CCl 4), methane (CH 4), ethylene (C2H 4), acetylene (C 2H 2), benzene (C 6H 6), acetic acid (CH 3COOH), and their derivatives. The atmosphere of Mars contains 96.2% CO 2. While it has been thought that silicon might take the place of carbon in forming a host of similar compounds, it is now not possible to form stable compounds with very long chains of silicon atoms. Without carbon, the basis for life would be impossible. There are close to ten million known carbon compounds, many thousands of which are vital to organic and life processes. With hydrogen, oxygen, nitrogen, and other elements, it forms a very large number of compounds, carbon atom often being linked to another carbon atom. Coal, petroleum, and natural gas are chiefly hydrocarbons.Ĭarbon is unique among the elements in the vast number and variety of compounds it can form. It is a component of great rock masses in the form of carbonates of calcium (limestone), magnesium, and iron. In combination, carbon is found as carbon dioxide in the atmosphere of the earth and dissolved in all natural waters. Little information is presently available about this allotrope. "White" carbon is a transparent birefringent material. The interplanar spacings of "white" carbon are identical to those of carbon form noted in the graphite gneiss from the Ries (meteroritic) Crater of Germany. Under free-vaporization conditions above ~2550°K, "white" carbon forms as small transparent crystals on the edges of the planes of graphite. In 1969 a new allotropic form of carbon was produced during the sublimation of pyrolytic graphite at low pressures. The hexagonal alpha type can be converted to the beta by mechanical treatment, and the beta form reverts to the alpha on heating it above 1000☌. Naturally occurring graphites are reported to contain as much as 30% of the rhombohedral (beta) form, whereas synthetic materials contain only the alpha form. These have identical physical properties, except for their crystal structure. Graphite exists in two forms: alpha and beta. Ceraphite is one of the softest known materials while diamond is one of the hardest. A fourth form, known as "white" carbon, is now thought to exist. FormsĬarbon is found free in nature in three allotropic forms: graphite, diamond, and fullerines. The energy of the sun and stars can be attributed at least in part to the well-known carbon-nitrogen cycle. About 30% of all industrial diamonds used in the U.S. Diamonds are now also being recovered from the ocean floor off the Cape of Good Hope. Natural diamonds are found in kimberlite of ancient volcanic "pipes," found in South Africa, Arkansas, and elsewhere. Carbon in the form of microscopic diamonds is found in some meteorites. 
It is found in abundance in the sun, stars, comets, and atmospheres of most planets. Carbon, an element of prehistoric discovery, is very widely distributed in nature. Carbon fiber is extremely strong and is used as a structural material when both strength and light weight are required. A drawing of a carbon image, and carbon fiber. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
